Fixation stability in cementless hip revision arthroplasty
Project description
Initial stability of cementless femoral stems after hip revision arthroplasty is crucial to restore hip biomechanics and ensure durable fixation. Various implant designs utilize different features to improve the axial and rotational stability in patients with proximal femoral defects. This study aimed to develop an experimental-computational framework for evaluating the fixation stability of novel cementless hip revision implants during activities of daily living.
Two cementless revision femoral stems were currently evaluated: RECLAIM Modular is a modular femoral system with a press-fit, splined, and tapered distal stem, RECLAIM Monobloc is a monobloc version of the same implant with additional splines between the primary splines, referred to as RECLAIM Advanced Splines (RAS), for enhancing cortical contact (Fig.1a). Each leg of a cadaveric lower limb specimen was broached to size for a primary implant to simulate a femoral defect, reamed, then implanted with RECLAIM Modular on one side and RECLAIM Monobloc on the contralateral side. The denuded femurs were potted into fixtures and mounted into an AMTI VIVO to simulate gait and stair descent (Fig.1b). Experimental boundary conditions were derived from the OrthoLoad database (force-control for anterior-posterior, medial-lateral, and superior-inferior degrees of freedom). Relative displacements between the proximal exposed implant-bone interface were recorded using a digital image correlation system.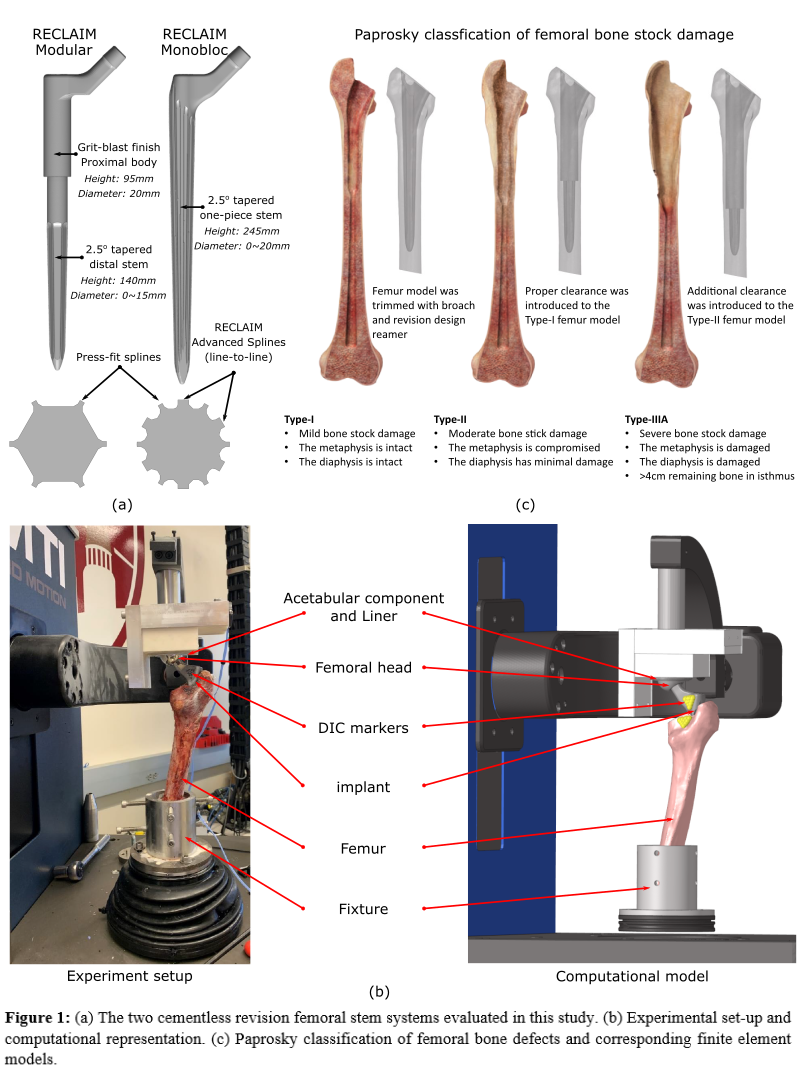
Finite element models of the specimen were developed in Abaqus/Standard from computed tomography (CT) scans (Fig.1b). Implant alignments were reconstructed using surface models of the implant and bone obtained from optical scans. The models were loaded with the boundary conditions from the experiment. The proximal implant-bone relative displacements were predicted and compared with the experimental measurements. After validation, virtual defects were added to the bone models to simulate different levels of bone loss (Paprosky Classifications Type-I, -II, and -IIIA) (Fig.1c). The two designs were then implanted into the same femur models and evaluated using the same activity loading. Implant-bone interface micromotions were investigated and compared. The models were additionally tested by applying a 10Nm torque and 1500N axial force to the implants to evaluate the axial and rotational stability of the two designs, respectively. The corresponding implant rotation/subsidence and maximum bone strain energy density (SED) across the femurs were compared. Uncertainty bounds for the predictions were generated by perturbing the interference fit on the primary splines from 0-100µm.
Acknowledgements
Supported by DePuy Synthes Products, Inc
References
- Weiss, R. J., et al., (2011). Journal of Arthroplasty, 26(1):16-23
- Kinkel, S., et al., (2013). International Orthopaedics, 37(10):1877-1883
- Della Valle C. J., et al., (2004). Clinical Orthopaedics and Related Research, 420: 55-62
- McCarthy, J. C, et al., (1997). Clinical Orthopaedics and Related Research, 344: 162-171
